
ELISA Kits
- • Anemia ELISA kits
- • Allergy ELISA kits
- • Autoimmune Disease kits
- • Bone Metabolism ELISA kits
- • Blood bank ELISA kits
- • Cancer ELISA kits
- • Cardiac Markers ELISA kits
- • Diabetes Assays ELISA kits
- • Drug test ELISA kits
- • Fertility ELISA kits
- • Food ELISA kits
- • Infectious Disease ELISA kits
- • Other ELISA Kits
- • Parasitology ELISA kits
- • Steroid ELISA kits
- • Thyroid ELISA kits
Rapid Tests
- • Allergy Rapid tests
- • Bone Metabolism
- • Cancer Rapid tests
- • Cardiac markers Rapid tests
- • Drug Tests
- • Fertility Rapid tests
- • Hepatitis Panel
- • Infectious Disease & other tests
- • Other
- • Ovulation Rapid tests
- • Pregnancy tests
- • Urine Reagent Strips tests
IFA Kits
Chemiluminescence Immuno Assays
- • Allergy Assays
- • Autoimmune Thyroid Assays
- • Cardio-Vascular Monitoring
- • Diabetes Assays
- • Fertility Assays
- • Growth Deficiency
- • Infectious Disease Assays
- • Others
- • Steroid Assays
- • Thyroid Assays
- • Tumor Marker Assays
Serology Tests
- • ASO (Anti-Streptolysin-O)
- • CRP (C-Reactive Protein)
- • Mono (Infectious Mononucleosis)
- • RF (Rheumatoid Factor)
- • RPR (Rapid Plasma Reagin)
- • SLE (Systemic Lupus Erythematosus)
Instrumentation



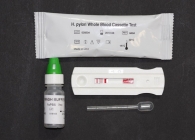
H. Pylori Rapid Test (Serum & Whole Blood )
| Name |
H. Pylori Rapid Test Kit |
|---|---|
| Full name |
Human Helicobacter Pylori Rapid Test RapiCardTM InstaTest (Cassette, Serum & Whole Blood ) |
| Category Name | Infectious Disease & other tests |
| Test | Bulk or Boxed in 25 or 50 tests |
| Specificity | 94.1 % |
| Sensitivity | 95.1% |
 |
 |
 |
H. Pylori Rapid Test (Serum & Whole Blood ) description:
The One Step H. pylori Serum and whole blood Rapid Test is a rapid lateral flow, qualitative immunoassay. It is intended for use at point of care facilities to detect the presence of IgG antibodies specific to Helicobacter pylori (H. pylori) in human blood or serum. It provides an aid in the diagnosis of infection by H. pylori.
Materials Provided with H. Pylori Rapid Test kit:
1. 25 H. Pylori Rapid Test devices
each sealed in a pouch with a dropper pipette
2. Wash buffer, 7 ml
3. Instruction for use
Materials required but not provided:
1. Lancet or other blood collection device
2. Clock or timer
H. Pylori Rapid Test kit Background Information
Helicobacter pylori has been associated with a variety of gastrointestinal diseases including gastritis, duodenal and gastric ulcer, non-ulcer dyspepsia, gastric adenocarcinoma and lymphoma. The exact role that H. pylori plays in gastrointestinal disease still needs to be precisely defined and is the subject of ongoing research. However, the prevalence rates for H. pylori infection as demonstrated by histological and bacteriological methods can approach 90% in patients who present clinical symptoms of the gastrointestinal diseases. H. pylori does not appear to invade the bloodstream since no isolates yet have been detected using commercial blood culture methods. H. pylori infections occur in human populations throughout the world. In developed countries, about 50% of the population may have H. pylori infection by the age of 60 years, while only 10-20% of adults in the third decade of life have it. In patients who present clinical symptoms relating to the gastrointestinal tract there are two major methods of investigation: invasive and noninvasive. Invasive methods include culture of gastric biopsy samples, histologic examination of stained biopsy specimens, or direct detection of the urease activity in the biopsy (CLO test). Noninvasive techniques include urea breath tests and serological methods. Serologic tests are employed to detect antibodies as human immune response to H. pylori. Two methods appear to be of great interest regarding their use in H. pylori routine serology, namely the ELISA and the Western immunoblot because they offer the most versatility in regards to immunoglobulin specificity and relative ease of use. This H. pylori Rapid Test detects IgG antibodies specific to H. pylori infection in patient blood or serum. It is a noninvasive method and does not use radioactive isotopes; the assay procedures are easy and do not require professional training; it provides a rapid result. It is a useful on-site aid in the diagnosis of H. pylori infection.
H. Pylori Rapid Test Principle
This assay is a double antigen chromatographic lateral flow immunoassay. The test strip in the device includes a burgundy-colored conjugate pad containing colloidal gold coupled with H. pylori antigens and nitrocellulose membrane containing a test line (T line) and a control line (C line). The T line is coated with H. pylori antigens, and the C line is coated with goat anti-H. pylori antibody. The antigens used in this device are from H. pylori cell lysate. When IgG antibodies specific to H. pylori are present in the specimen, the T line will become a burgundy-colored band. If antibodies to H. pylori are not present or are present below the detectable level, no T line will develop. The C line should always appear as a burgundy-colored band regardless of the presence of antibodies to H. pylori. The C line serves as an internal qualitative control of the test system to indicate that an adequate volume of specimen has been applied and the flow occurred. For additional details please refer to the instructions for use.
Notable features of H. pylori Rapid Test:
-Reliable and inexpensive
-Convenient and easy-to-understand directions
-Clear preparation procedures
-Simple and safe specimen collection directions
-Comprehensive package of required materials and components
-Rapid and accurate test results
See H. pylori Rapid Test Product Insert for specific details on preparation, procedures, quality control, and interpretation of test results.
H. Pylori Rapid Test Results Interpretation
The result is positive if both the C line and T line appear, indicating that the IgG antibodies specific to H. pylori are detected. If only the C line appears in the control region, the test indicates that no antibodies to H. pylori are detected and the result is negative. For additional details please refer to the instructions for use.
H. Pylori Rapid Test Kit Performance Characteristics
Sensitivity and Specificity:
This device was evaluated with 296 confirmed clinical serum specimens, 144 were positive and 152 were negative. All the specimens were blind labeled. The sensitivity of this device was calculated to be 95.1% (137/144) and the specificity to be 94.1% (143/152).
Cross Reactivity:
Other closely related microorganisms were evaluated for cross reactivity with the test. Proteins of those microorganisms were spiked into the H. pylori positive and negative specimens at a high
concentration and tested separately. None of the microorganisms affected the test results positive or negative.
Interference:
No interference was found with Common Biological Analytes. For additional details please refer to the instructions for use.
Diagnostic Automation Inc. also offers other Infectious Diseases Rapid tests, and Infectious Diseases ELISA test kits. For more information about Rapid tests, CLIA tests, IFA kits, or ELISA test kits, please contact our Customer Service Representatives at 818-591-3030 .
H. Pylori Related Rapid Test Kits:
H. Pylori Rapid Test (Serum Cassette) 118561-1
http://www.rapidtest.com/index.php?i=H.-Pylori-Serum-Rapid-Test-&id=387&cat=26
H. Pylori Antigen Rapid Test (Cassette) 118564-1
http://www.rapidtest.com/index.php?i=H.-Pylori-Antigen-Rapid-Test-(Cassette)-&id=388&cat=26
H. pylori ELISA Kits
H. pylori IgG ELISA kit
http://www.rapidtest.com/index.php?i=H.-pylori-ELISA-Test&id=140&cat=16
H. pylori IgM ELISA kit
http://www.rapidtest.com/index.php?i=H.-pylori-IgM-ELISA-kit&id=142&cat=16
H. Pylori Antigen ELISA kit
http://www.rapidtest.com/index.php?i=H.-Pylori-Antigen-ELISA-kit&id=613&cat=16
Additional H. pylori kits available are:
H. pylori IgA CLIA, H. pylori IgG CLIA & H. pylori IgM CLIA Kit
H. pylori CLIA Kits
http://www.rapidtest.com/index.php?product=Infectious-Disease-Assays&cat=39
Diagnostic Automation Inc. also provides other Rapid Test Kits:
http://www.rapidtest.com/index.php?product=Rapid-Tests
For more information about Rapid Tests, ELISA Kits, IFA Kits, CLIA Test Kits, or Serology tests, please see our website home page, or contact our Customer Service Representative
at 818-591-3030.
Product Note:
For this H. pylori Rapid Test, any sediment in serum specimens should be removed by centrifugation. Avoid using any turbid specimens, which may be contaminated by microorganisms.
ELISA kits - Rapid tests- Drug tests- Pregnancy test - IFA kits - CLIA assays - Serology tests - Instrumentation
©1992 Diagnostic Automation/Cortez Diagnostics Inc. All rights reserved.


 H. Pylori Serum Rapid Test
H. Pylori Serum Rapid Test
 Dengue Rapid Test
Dengue Rapid Test
 Chlamydia Rapid Test (Female)
Chlamydia Rapid Test (Female)
 Malaria Rapid Test
Malaria Rapid Test
.jpg&w=195&h=140) Syphilis Rapid Test
Syphilis Rapid Test 








